-
New course available

Learn how the EU Regulation coming in 2025 will affect HTA processes and how you can get involved.
-
Train with us

Interested in an online training session on EU HTAR? Click the link to pre-register!
-
Learn all about HTA

Follow all our Health Technology Assessment courses. Content is continually updated throughout the HTA4Patients project.
HTAR in a Nutshell
Are you curious to learn more about the EU Health Technology Assessment Regulation (HTAR)?
In this video, we’ll cover the key changes introduced by the HTAR regulation, its objectives and purposes, and what it means for Health Technology Assessments (HTAs) in the EU.
We’ll also explore the stakeholders involved, their primary roles, and how patients and patient organizations can engage and contribute to this important process.

What is HTA?
According to Health Technology Assessment International (HTAi.org) Health Technology Assessment (HTA) is a multidisciplinary process that uses explicit methods to determine the value of a health technology at different points in its lifecycle. The purpose is to inform decision-making in order to promote an equitable, efficient and high-quality health system.
About HTA4Patients Project
The European Patients Academy on Therapeutic Innovation (EUPATI) is leading a 3-year training project entitled ‘HTA4Patients’ from 01 March 2023.
The European Union’s new framework for Health Technology Assessment (HTA) regulation (EU 2021/2282) represents a unique opportunity to strengthen the involvement of patient communities in HTA processes. Strong and informed input from patients and patient representation is necessary to support this new framework and efficient HTA.
HTA4Patients will enhance the education, training and information that EUPATI already provides on HTA via its Open Classroom and Toolbox empowering patients and patient organisations to play a vital role in the implementation of the new framework.
-
![]()
Objective 1
Creating an e-learning training course on the new HTA Regulation and what it means for patients
![]()
Objective 2
Updating EUPATI’s existing patient training content on HTA aligning with the new EU regulation
![]()
Objective 3
Developing interactive online training sessions (in 5 languages). Preparing participants taking part in joint clinical assessments and joint scientific consultations with national HTA bodies
![]()
Objective 4
Implementing sustainability measures to ensure continuity of the project outputs in the future
Our work plan
WorkPackages 1-4
-

WP 1
- Project Management
- Coordination
- Infrastructure
-

WP 2
- E-learning training course
-

WP 3
- Online training sessions
-

WP 4
- Communication
- Dissemination
- Outreach and Sustainability
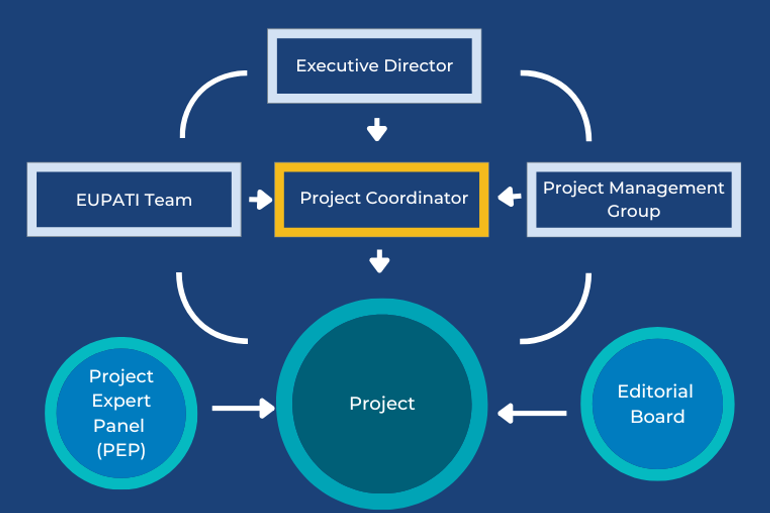
Project Governance
Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or European Health and Digital Executive Agency (HADEA). Neither the European Union nor the granting authority can be held responsible for them.
More information















